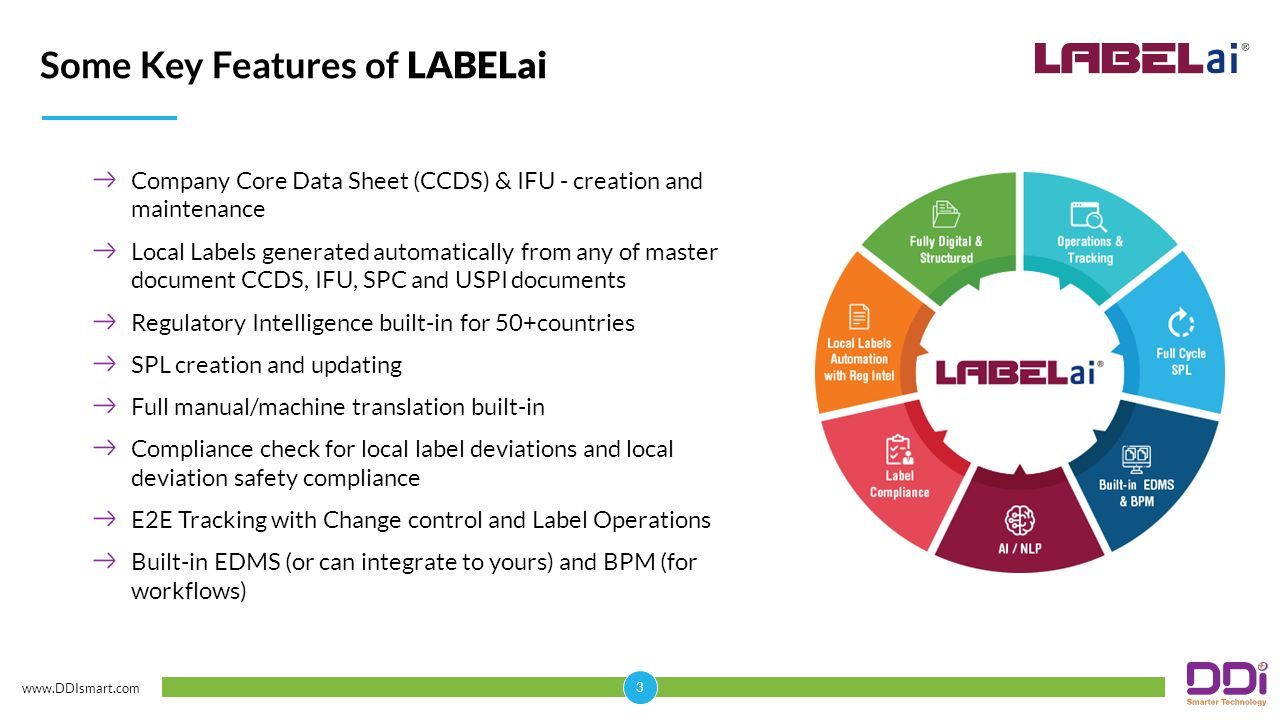
Fillable Online Procedural guidance on inclusion of declared interests in the European Medicines Agency s electronic declaration of interests form (for scientific committees members and experts). EMA/627294/2014 Fax Email Print - pdfFiller
Company Core Data Sheets & Core Safety Information April 21 – 22, 2014 Hyatt Arlington ~ Arlington, Virginia